Structure of a Cell
Biology 372 looks at cellular contractility.
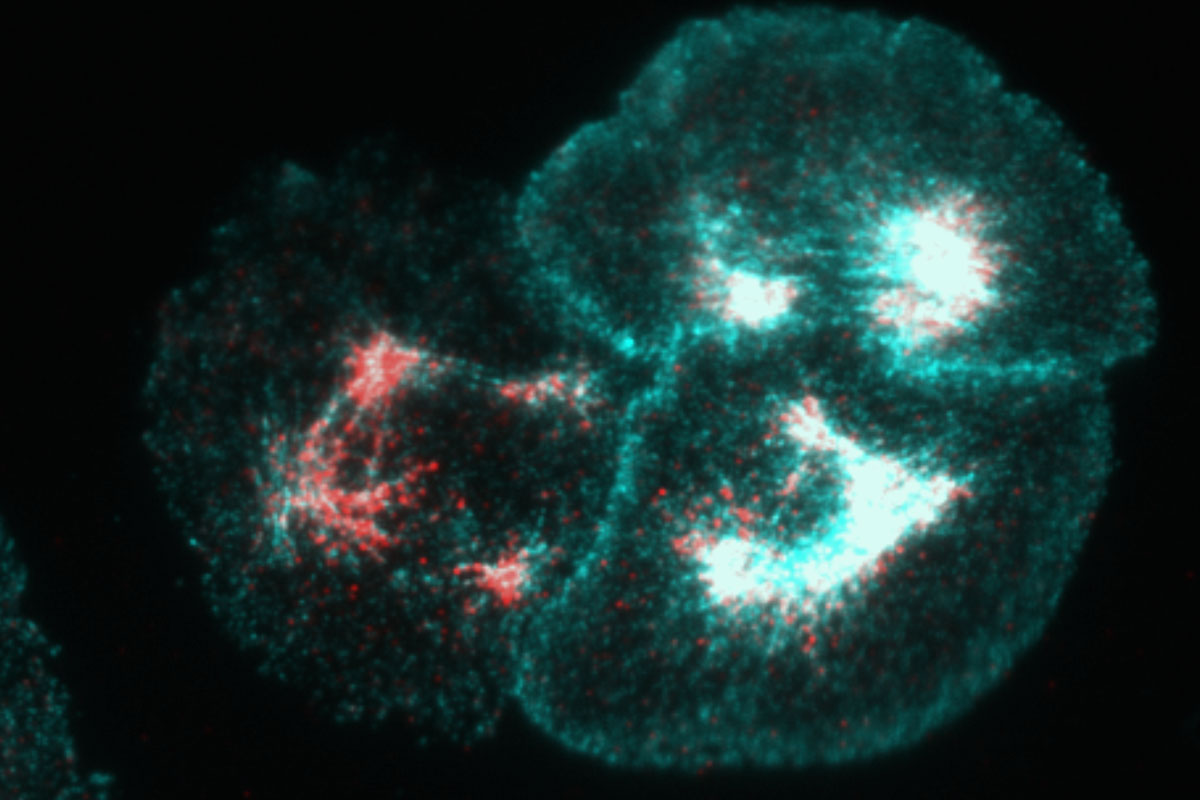
This image shows three Drosophila S2R+ cells (cells cultured from fruit fly embryos) stained with actin in cyan and phosphorylated non-muscle myosin II in red. Actin and non-muscle myosin II are proteins that work together to generate cellular contractility, which is critical to morphogenesis—the process by which cells change shape.
Morphogenesis is the driving force behind the formation of our gut, the tube that eventually becomes our spine, and the folding of tissue that creates our organs and ducts. It is essential to all multicellular life forms; without it we would never have evolved from one-cell organisms.
Non-muscle myosin II (which is closely related to skeletal and cardiac myosin and more than likely came before these specialized types of muscle myosins) is regulated by phosphorylation, a post-translation modification that in this case turns it “on.” Furthermore, non-muscle myosin II is also a motor protein, which means it “walks” along actin filaments, and, in doing so, causes these filaments to slide past one another. This sliding leads to contractility. Students in my Biology 372: Cell Biology course, as a part of the lab portion of the class, participate in a National Science Foundation funded project initiated by Prof. Anna Ritz and myself where they are using both computational and wet lab methods to identify novel regulators of this cellular contractility.
Tags: Academics, Object of Study, Professors, Research