Molecular Seduction
A look into Chemistry 391: Structural Biochemistry
All chemists are beset by the same hobgoblin: the need to visualize the molecular structures they work with. In the old days, they built models out of balls and sticks. Today, of course, they use computers. The resulting models are gorgeous—but deceptive.
“Models themselves are inherently fragile,” says Prof. Arthur Glasfeld [chemistry 1989–], because they represent only one possible way of interpreting imperfect experimental data. So how can you gain insight from a model without being seduced by it?
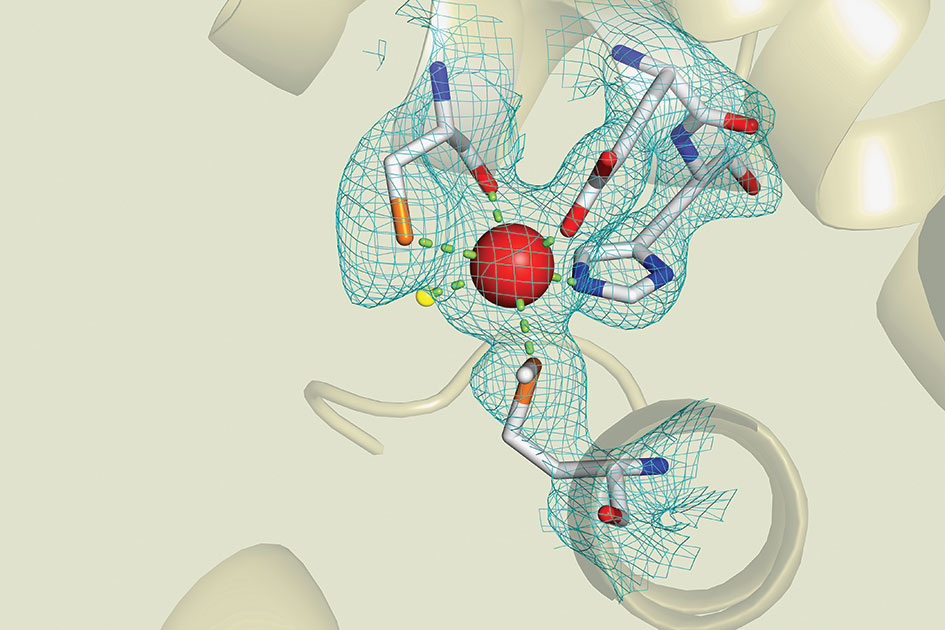
In his Structural Biochemistry class, Prof. Glasfeld and his students examine this model of the iron-dependent regulator (IdeR) protein extracted from the deadly tuberculosis mycobacterium.
First published in 1998, the model shows a central zinc ion (the red sphere) connected to arms composed of carbon (white), nitrogen (blue), sulphur (orange), and oxygen (red). Beyond the frame of focus, the protein chain coils into spiralling ribbons. A lone molecule of water (yellow) peeks out on the lefthand side.
But this model is only an approximation—the arms may exist anywhere inside the blue mesh indicating the zone of uncertainty. Glasfeld then challenges the students to refine and improve the model, based on current data.
This is more than a theoretical exercise. IdeR is an essential part of the germ’s ability to elude the human immune system through its interaction with metal ions. The better we understand it, the better our chances of finding ways to defeat it.
Tags: Professors, Academics, Object of Study