|
|

|
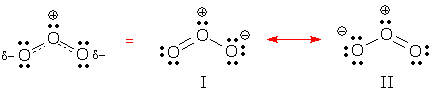
Major vs. minor resonance contributors Ozone is a symmetric molecule, so I and II contribute equally to our picture of this molecule.
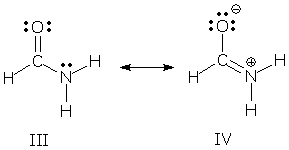
Most resonance hybrids are not symmetric, however, and their contributors do not play equal roles. Formamide is a superposition of III and IV, but its structure and properties more closely resemble III: the atoms carry only small charges, the CO bond is closer to a double bond, and the CN bond is closer to a single bond. In this case, we say III is the major contributor and IV is the minor contributor.
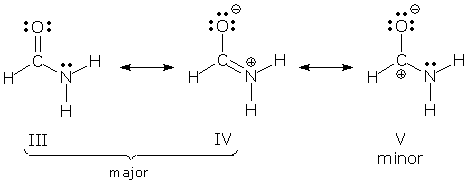
Ambiguous terminology. "Major" and "minor" are still used when a molecule is a superposition of three or more contributors. In these situations, "major" can be applied to all of the larger contributors, and "minor" to all of the lesser contributors. For example, formamide is sometimes viewed as a superposition of three contributors:
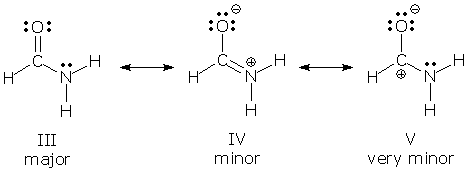
V plays a much smaller role than III or IV, so we might refer to V as a minor contributor, and III and IV as the major contributors. Sometimes, though, we want to keep the relative roles of all three contributors clear. In this case, we use the following terminology:
Ranking contributors. There are several ways to identify major, minor, and very minor contributors:
|