THE IMMORTAL JELLYFISH
GEOFF DERVEN / BIOLOGY 342 / FALL 2010
| HOME | PHYLOGENY | ONTOGENY | MECHANISM | ADAPTIVE | REFERENCES | MORE? |
MECHANISM
Mechanism is the study of the “material” workings of an animal’s behavior. Through examining the hormonal, neuronal, physiological, and otherwise somatic factors that lead to an organism’s particular condition, we can grasp the most literal understanding of how an action manages to “work.” Just the same, mechanism is not entirely a question of bodily logistics, but also incorporates an animal’s environmental context – rather, how externalized inputs may initiate a particular response. Here we do not consider how something came to be, nor to what end it serves, but rather how the behavioral process itself actually occurs.
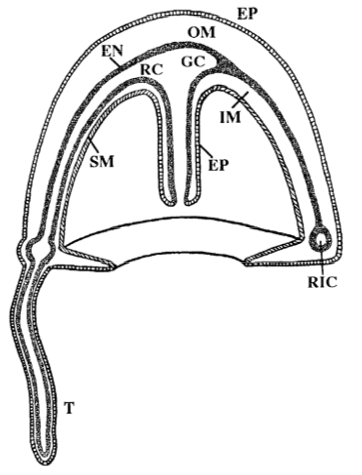
An anatomized T. nutricula medusa: EP= epidermis, OM = outer mesogloea, EN = endoderm, IM = inner mesogloea, SM = striated muscle, GC = gastral cavity, RC = radial canals, RIC = ring canal, T = tentacle (Piraino, et al., 1996).
T. nutricula do not revert their life cycle spontaneously, but instead initiate the process in response to physical stress, subject to the likes of starvation or tissue damage. Once they experience such bodily traumas, they begin to transdifferentiate, returning to their earliest polyp form (Miglietta & Lessios, 2009; Aerne, 1996). The umbrella of the medusa stage is then inverted, absorbing its tentacles into its interior. Stolons are formed, after which – as two days pass – polyps begin to differentiate, before finally culminating in a “new” hydroid colony (Hongbao Ma, 2010). Piraino, et al., (1996) observed a 100% success rate in this regeneration, thereby concluding that an organismic death simply does not occur for the species.
A call for further molecular research on the T. nutricula was only first expressed in the early 1990s, as its unusual, life-reversing behavior first came to light (Bavestrello, et al., 1992). Transdifferentiation itself is also a more recent scientific discovery, previously understood as “metaplasia,” and later, “cell transformation” (Piraino, et al., 1996). Determining what mechanisms are really at work in this regenerative process has, thus far, proven to be unsuccessful – a great deal of uncertainty remains. Further still, as transdifferentiation is typically limited to the subsets of an organism (versus T. nutricula’s virile, organismic plasticity), the mechanisms responsible for a totalizing cellular reversal are likely to be even more nuanced and interwoven in their operation.
In 1996, Piraino, et al. discussed how interstitial cells – arising from stem cells – along with numerous processes of DNA replication, may contribute to (or even be responsible for) transdifferentiation. Stem cells, however, are mostly limited to the characteristics of their parent generation; they only provide the slightest room for less specific differentation as they become particularized into maturity (Collas & Håkelien, 2003).
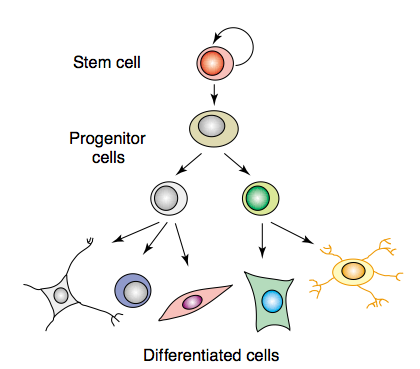
The maturation of cells also particularizes their function (Collas & Håkelien, 2003).
Transdifferentiation, then, is something seemingly altogether different, as it allows for already differentiated cells to switch to entirely new functions. As T. nutricula medusae devolve into polyps, their particular tissues have to be dissolved, regenerated, reorganized, and – most importantly – reprogrammed for a new function. This process could not simply occur by de-differentiating, but instead through a deeper sort of metamorphosis (Collas & Håkelien, 2003; Piraino, et al., 1996), transforming the very cells themselves.
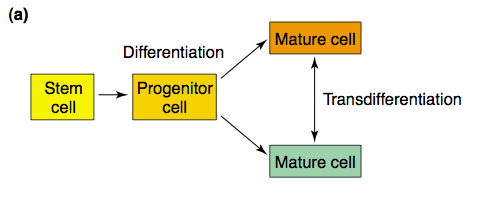
The non-alike processes of cellular differentiation and transdifferentiation (Collas & Håkelien, 2003).
To better pursue such complexities as these, Piraino, et al. (1996) isolated various components of T. nutricula medusae. They sought to localize the mechanism (or mechanisms) through which the medusae’s restorative potential is enacted, dissecting the organism into fragments. The cultured results suggest that the presence of various cells of the exumbrella and the gastrovascular system are necessary for its transformative processes to occur. But what is particular about these cells – and what exactly is required of them – remains unclear (Piraino, et al., 1996).
Once the most foundational aspects of these mechanistic hurdles are solved, further research may allow for us to understand the furthest depths of transdifferentiation – perhaps even learning to train our own cells. If the lasting stability of transdifferentiated cells proves to be sufficient, we may eventually see such a process become a vital player in the search for cures for cancer or diabetes, providing potential breakthroughs in the ever-growing field of cell therapy (Collas & Håkelien, 2003).