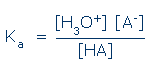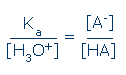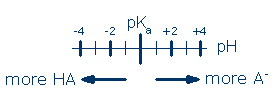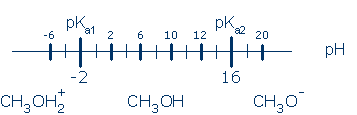
|
|

|
Predict a Molecule's Ionization State Background A molecule's charge changes whenever it gains or loses a proton, H+. The molecule's charge is known as its ionization state. The molecules of a substance can exist in different ionization states simultaneously. Consider the following reaction between an organic acid, HA, and water:
When this solution reaches equilibrium, molecules of the acid will be found in two ionization states, A- and HA, and molecules of water will be found in three, H3O+ , H2O, and HO- (not shown). If the mixture is dilute, the equilibrium concentrations of these species will be given by:
Ka(HA) is known as the acid dissociation constant of HA. This constant describes the intrinsic reactivity of HA towards water.
Predicting the ionization state of a simple acid, HA Before you can to predict an acid's ionization state, you must obtain Ka(HA) and the equilibrium concentration of H3O+ (or pH). The equilibrium ratio of HA and A- is described by the following equation (compare this equation to the one shown above):
From this equation, you can see that 50% of the acid molecules will be ionized when [H3O+] = Ka. It is more commonplace to have pKa and pH data. These data can be inserted into the previous equation after taking the antilog of each quantity. You can skip this step, however, by using the following chart:
Simply locate the pH of your solution relative to the pKa of your acid and read the dominant ionization state (note: the more protonated ionization state always lies on the lower pH side of pKa, and the less protonated state always lies on the higher pH side). You can also make quantitative predictions with this chart. Let's call "D" the distance from pH to pKa in pH units. The equilibrium ratio of [major ionization state]/[minor ionization state] = 10D. As an example, if the pH is 4 units higher than the pKa, the major ionization state is A- and [A-]/[HA] = 104. On the other hand, if the pH is 5 units lower than the pKa, the major ionization state is HA, and [HA]/[A-] = 105.
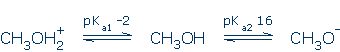
Predicting the ionization state of a polyprotic acid, H2A Suppose you have a compound that can enter into two acid-base equilibria, like methanol, CH3OH:
Once you know the pKa for each ionization, and the equilibrium pH, you can use the following chart to predict the major ionization state:
Notice that this chart is identical to the one in the previous section. the most protonated ionization state is the major species at very low pH. Then, as we move to the right (to higher pH), we remove a proton each time we pass a pKa. You still read the chart in the same way as before. Suppose the solution's pH is -4. This point lies to the left of pKa1 by 2 pH units, so most of the molecules will exist as CH3OH2+ (and the equilibrium ratio favoring this state is [CH3OH2+]/[CH3OH] = 102).
Review problems under construction |